Hop Latent Viroid detection in leafhoppers (Nephotettix cincticeps) feeding on Cannabis sativa sap
Kevin McKernan and Collin Palmer
Hop Latent Viroid detection in leafhoppers (Nephotettix cincticeps) feeding on Cannabis sativa sap
Kevin McKernan1 and Collin Palmer2
1)Medicinal Genomics
2)Vessel Life Sciences
Introduction
The cannabis field is experiencing its own pandemic related to a plant viroid that jumped from Hop (Humulus lupulus) to its close cousin Cannabis sativa. When and how this occurred is unknown but since the viroid sequence in Hop and Cannabis are identical it’s assumed it was a fairly recent introduction. The zoonotic leap from Hop to Cannabis was often assumed to be the result of various clandestine attempts to graft Cannabis flowers onto Hop and cross these plants to form a cannabinoid producing plant that look nothing like cannabis and thus potentially evade detection. This transmission assumption is based on the only (currently) known transmission route of HpLVd in cannabis being through mechanical transfer from cloning scissors. Cannabis can be easily cloned by cutting a branch off, dipping it in rooting hormone and planting it in rock wool. This cutting process can transfer HpLVd from a mother plant to all of its clones and to anything those scissors touch in the future.
However, various other viruses (LCV and BCTV) have been documented to make this leap through the tiny ‘scissors’ of insects feeding on plant sap.
Collin Palmer has been keeping HpLVd plants arounds to study them. After he noticed leafhoppers on his HpLVd infected plant, he decided to have them tested for HpLVd to see if they contain any viroid. His instincts were spot on.
Leafhoppers were collected from HpLVd infected plants and placed into a magnetic leaf lysis boiling solution.


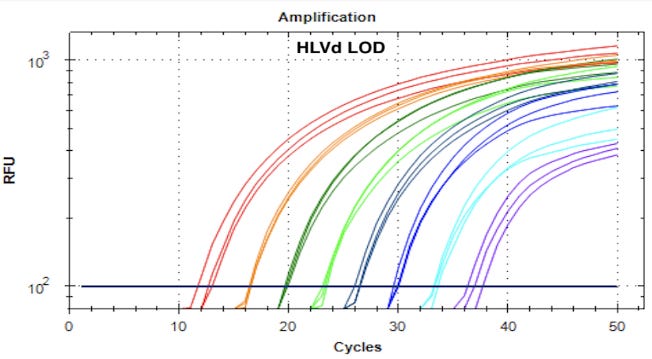
This magnetic boil is then removed, magnetically separated, and washed with 70% Ethanol. The DNA is eluted in water and run through a single copy sensitive RT-qPCR reaction.
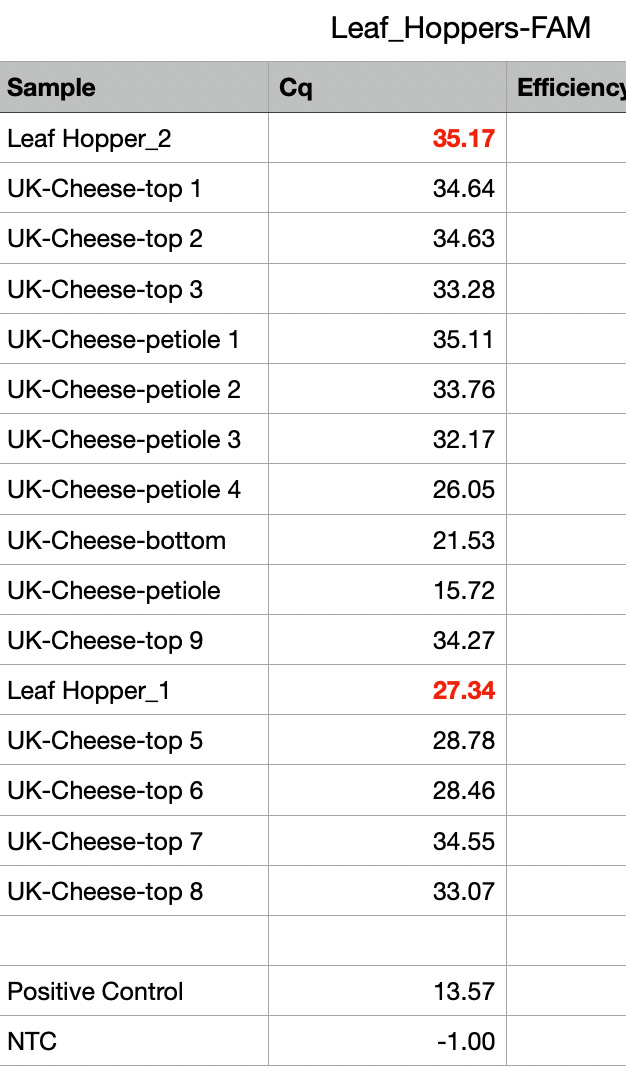
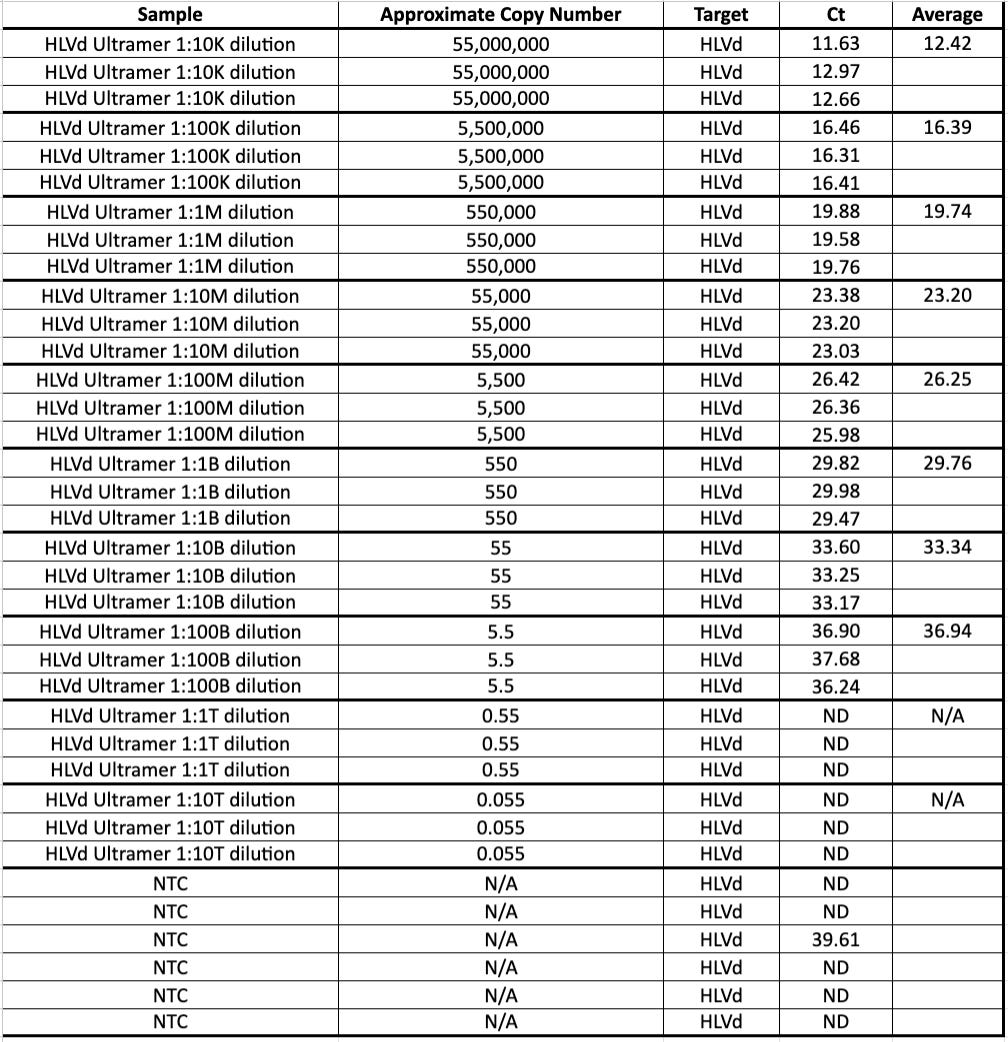
HpLVd CT =27.3 for leafhopper. This is more signal than many parts of the plant that are surveyed.
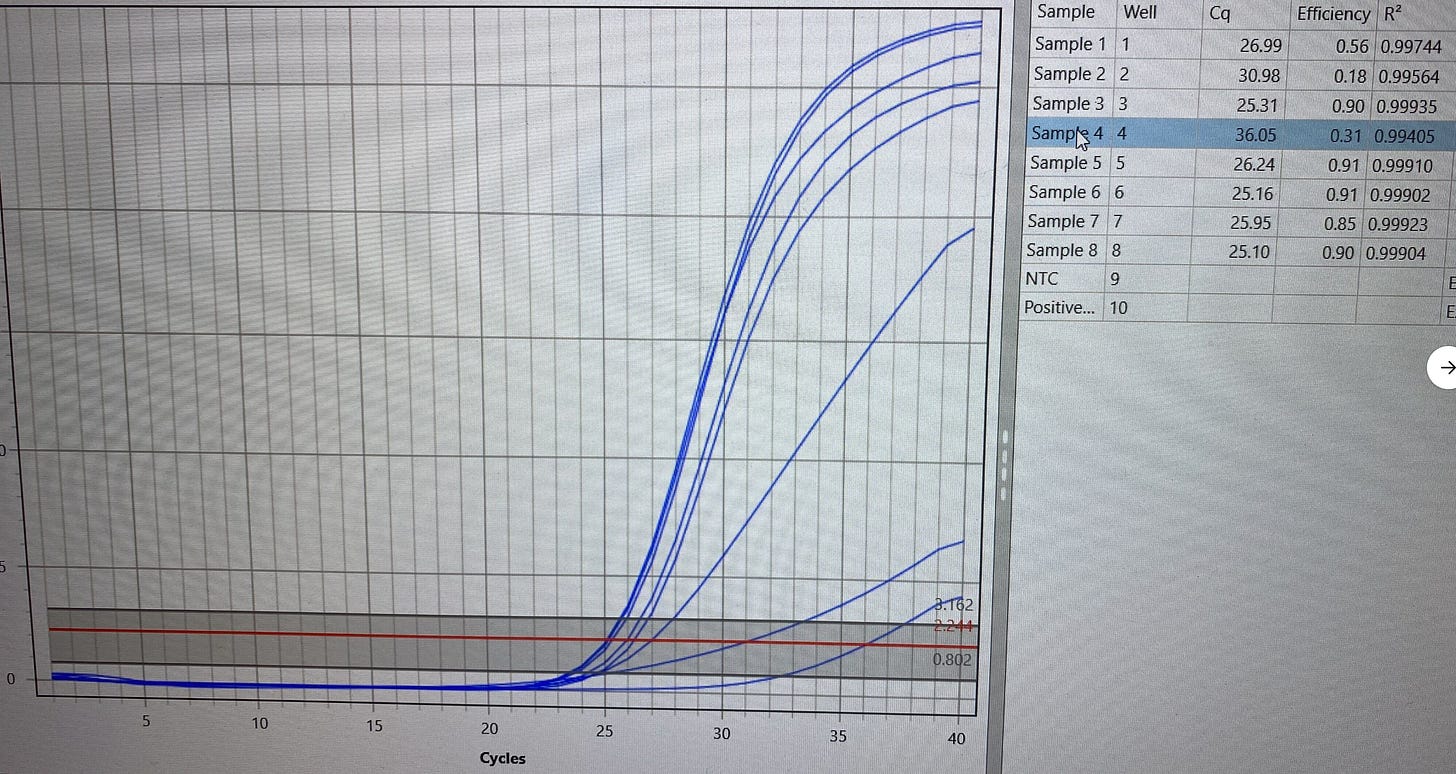
Concurrently, Cannabis RNA and DNA is also amplified as an internal control to prove the extraction and qPCR process is working properly.
The Cannabis Internal control is detected at a CT of 36. This is not surprisingly a very late CT. No NTC signal implies the cannabis signal from the insect is not background. Why is any cannabis RNA/DNA found in an insect? leafhopper are known to pierce the plant and feed on cannabis sap. This late Cannabis CT is believed to be a result of insects having ingested cannabis sap that is detectable upon lysis.
After HpLVd was detected in 2/2 leafhoppers, we collected 11 more leafhoppers from the infected UK-Cheese cultivar. We placed 5 of these leafhoppers in a bag with UK-Cheese leaves to feed on and 6 leafhoppers in a bag with a previously tested negative for HpLVd plant (Gary Payton) as a feed source. We left them at room temperature for 20 hours and then extracted DNA and RNA for qPCR with a simple magnetic bead boil prep.
Leafhoppers from HpLVd positive cultivar UK Cheese with 3 plant samples (right).
Leafhoppers from HpLVd negative cultivar Gary Payton with 2 plant samples (right).
Leafhoppers in MGC lysis reagent
Leafhoppers after boil

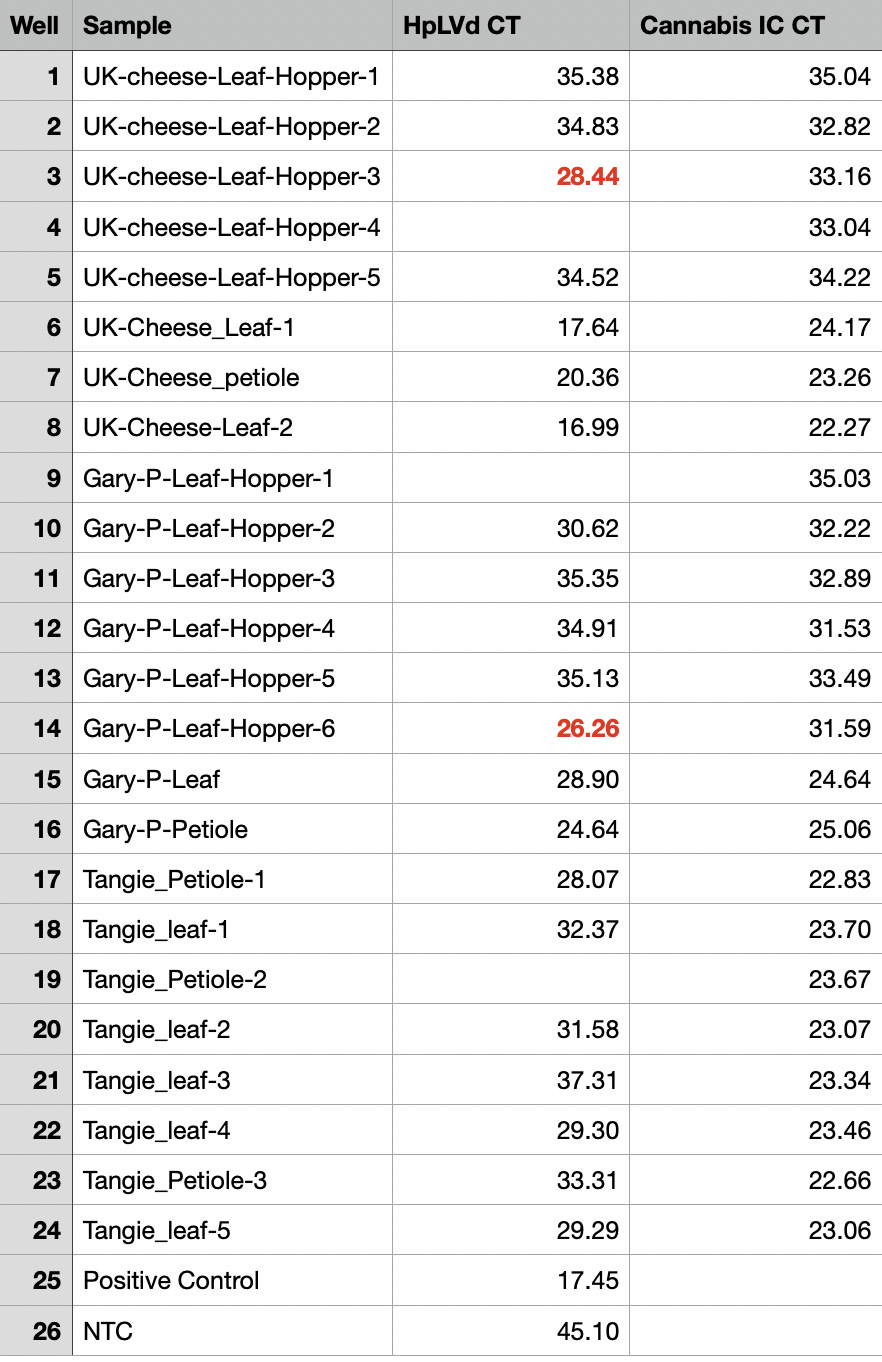
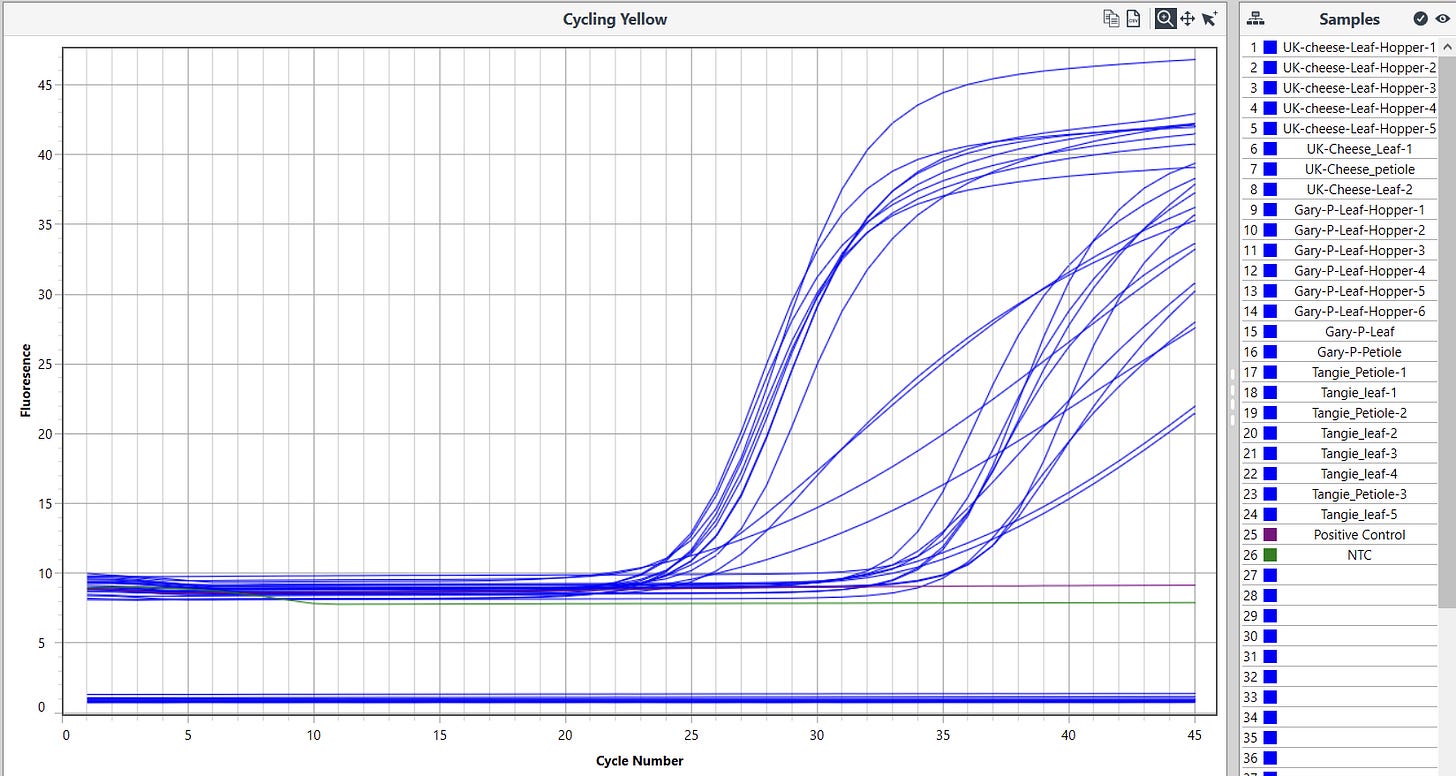
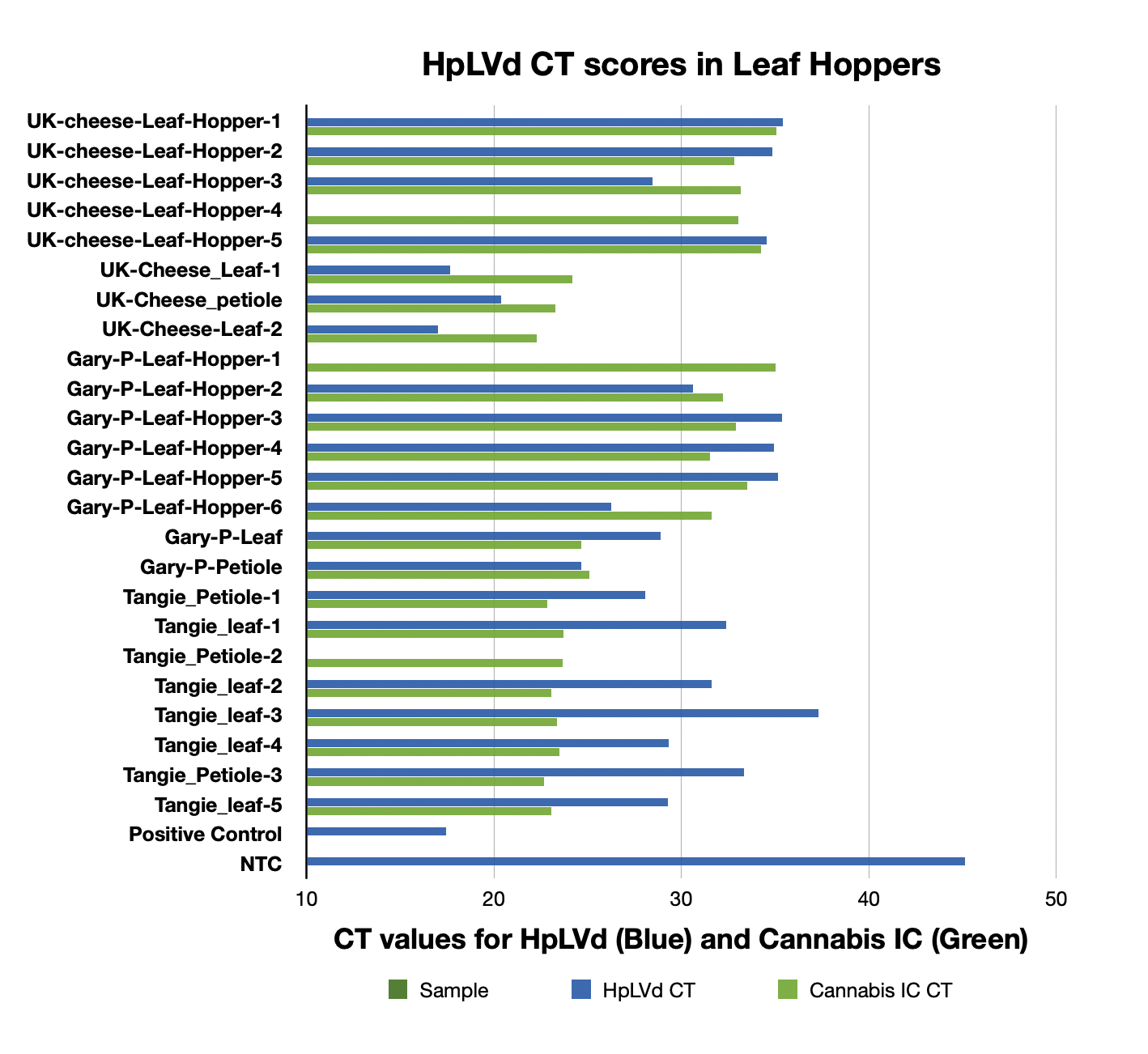
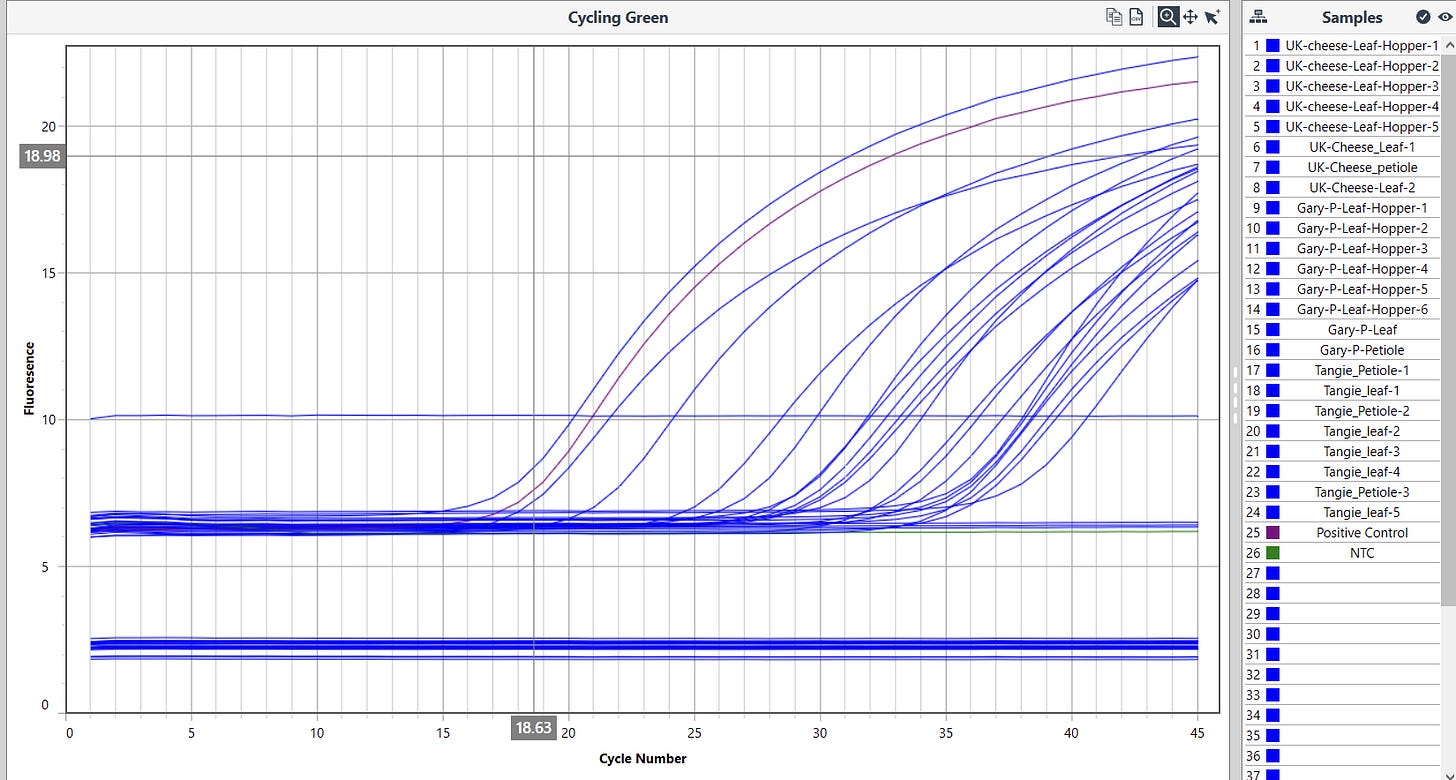
The expanded survey of 11 Leafhoppers reproduced the detection of HpLVd in 9/11 Leafhoppers. CT scores varied with 7/9 being late CT (n=9 average = CT 32.8). Two of leafhoppers in the second screen had CT scores of 26.3 and 28.4. The originally infected UK Cheese leaf samples had CT scores averaging 18.3 (n=3), while the cultivars that were negative in vegetative state (Gary Payton and Tangie) had much later HpLVd CTs scores (9,900 fold lower copy number or 13.3 delta CT) suggestive of later infection.
The Cannabis internal control amplicon was also detected in Leafhoppers but at a 1,000 fold lower levels than cannabis leaf samples tested. This is suggestive of detection of the Cannabis meal in the leafhoppers digestive track.
Methods
High sensitivity Lysis and RT-qPCR kits from Medicinal Genomics were used in this study according to the manufacturers protocols. 100µl of MGC HS-lysis buffer was used to boil leafhoppers and plant tissue (92C for 8 minutes). 75µl of lysates were purified with magnetic separation and eluted in 30µl ddH20. qPCR reactions were assembled using a MIC/MYRA automated liquid handler to avoid contamination and cycled 40-45 cycles.
Conclusions
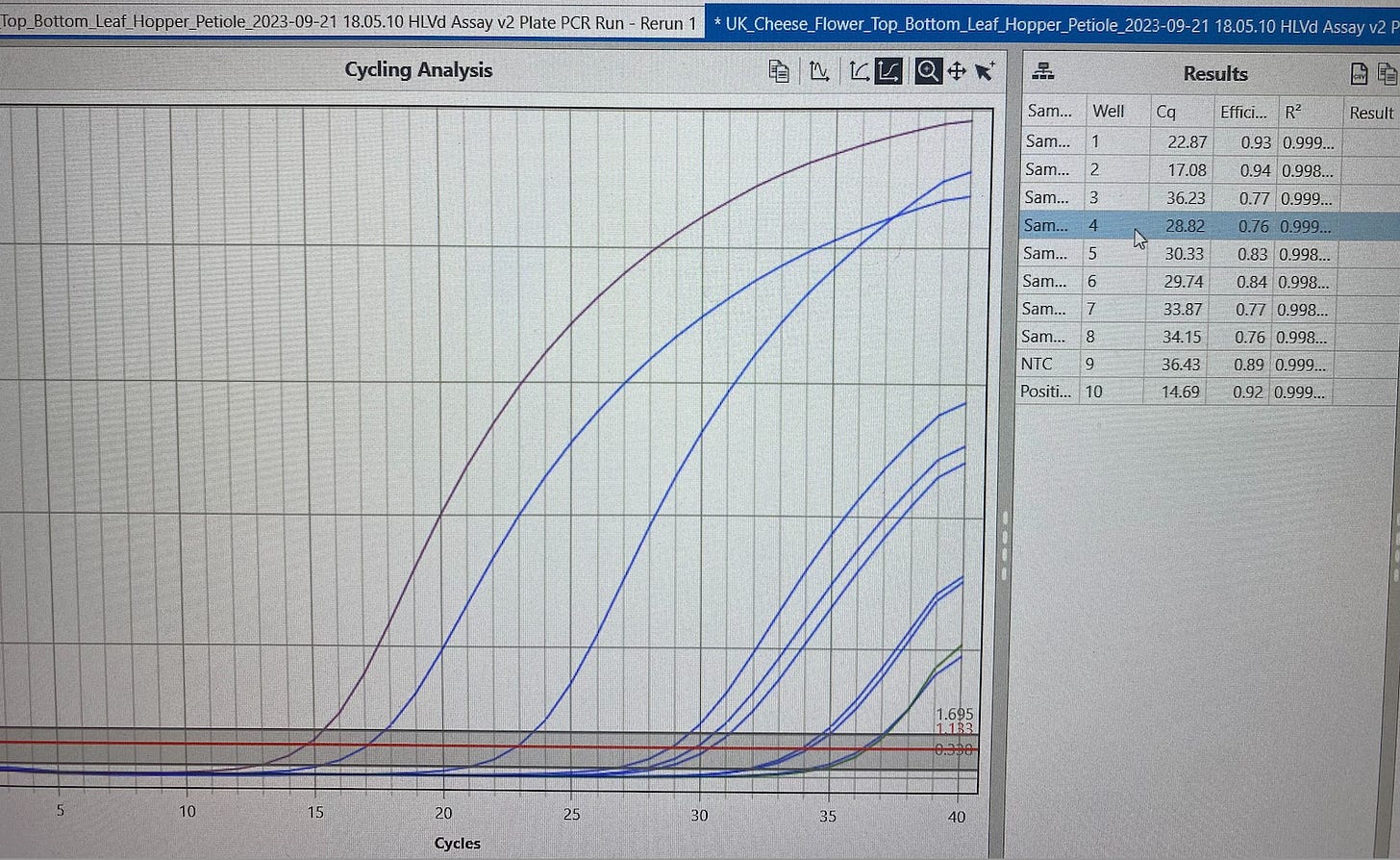
Leafhoppers feeding on HpLVd infected cannabis plants are PCR positive for the HpLVd RNA. Tangie plants local to infected plants (but not within root contact) have late CTs for HpLVd in flower. Gary Payton was in root contact reach of UK-Cheese but Tangie was not. More controlled work is required to confirm that the detected viroid load can in fact transmit to another uninfected plant through insect feeding alone. More insects need to be surveyed with more controlled plant isolation protocols. The speciation of the leafhoppers has not been confirmed genetically. Given the literature documentation of other viruses transmitting in this manner, growers should assume insects that feed on the plant are a potential vector for transmission of HpLVd.







Am I the only one reading this that finds it hilarious? Brilliant scientist doing highly technical work involving a plant that gets you high and is named after an NBA hall of famer. It's just a tad bit bizarre.
Kevin & Collin - "The zoonotic leap from Hop to Cannabis was often assumed to be the result of various clandestine attempts to graft Cannabis flowers onto Hop and cross these plants to form a cannabinoid producing plant that look nothing like cannabis and thus potentially evade detection."
Or to make beer that really gets you dazed & infused. From the evidence, man's wisdom must walk hand in hand with his idiocy.